STANDARD OPERATING PROCEDURES or SOP
What is Standard Operating Procedure? A Standard Operating Procedure (SOP) is a set of written instructions that document a routine or repetitive activity followed by an organization.The development and use of SOPs are an integral part of a successful quality system as it provides individuals with the information to perform a job properly, and facilitates (làm cho dễ dàng; làm cho thuận tiện) consistency (trước sau như một) in the quality and integrity (toàn vẹn, toàn bộ) of a product or end-result. The term “SOP” may not always be appropriate and terms such as protocols (bản dự thảo đầu tiên hoặc nguyên gốc), instructions, worksheets, and laboratory operating procedures may also be used. How can you tell if a document is a SOP, Procedure, or Work Instruction (OMS/Operational Method Sheet)? Also, what are the connections between each of these documents? Do you need to write an SOP before the OMS?
and facilitates (làm cho dễ dàng; làm cho thuận tiện) consistency (trước sau như một) in the quality and integrity (toàn vẹn, toàn bộ) of a product or end-result. The term “SOP” may not always be appropriate and terms such as protocols (bản dự thảo đầu tiên hoặc nguyên gốc), instructions, worksheets, and laboratory operating procedures may also be used. How can you tell if a document is a SOP, Procedure, or Work Instruction (OMS/Operational Method Sheet)? Also, what are the connections between each of these documents? Do you need to write an SOP before the OMS?
SOPs often including elements like design, manufacturing and distribution, a standard operating sheet will provide an estimate of the time required to complete each stage. It is a tool used in lean manufacturing that aims to improve the process as a whole through standardization and for training of staff. Standard Operating Procedures describe both technical and fundamental programmatic operational elements of an organization that would be managed under a Work Plan – a Quality Assurance – a Quality Assurance Projects Plans (EPA QA-R5) – a Quality Management Plan (QA/R-2) – and the EPA Quality Manual. These documents are designed to provide guidance in the preparation and use of an SOP within a quality system.
Purpose: SOPs detail the regularly recurring work processes that are to be conducted or followed within an organization. They document the way activities are to be performed to facilitate consistent conformance to technical and quality system requirements and to support data quality. They may describe, fundamental programmatic actions and technical actions such as analytical processes, and processes for maintaining, calibrating, and using equipment. SOPs are intended to be specific to the organization or facility whose activities are described and assist that organization to maintain their quality control and quality assurance processes and ensure compliance with governmental regulations. It minimizes opportunities for miscommunication and can address safety concerns. When historical data are being evaluated for current use, SOPs can also be valuable for reconstructing project activities when no other references are available.
SOPs detail the regularly recurring work processes that are to be conducted or followed within an organization. They document the way activities are to be performed to facilitate consistent conformance to technical and quality system requirements and to support data quality. They may describe, fundamental programmatic actions and technical actions such as analytical processes, and processes for maintaining, calibrating, and using equipment. SOPs are intended to be specific to the organization or facility whose activities are described and assist that organization to maintain their quality control and quality assurance processes and ensure compliance with governmental regulations. It minimizes opportunities for miscommunication and can address safety concerns. When historical data are being evaluated for current use, SOPs can also be valuable for reconstructing project activities when no other references are available.
WORKFLOW PROCESS: Create and initially define —» Review —» Revise —» Approve —» Publish.
Writing Standard Operating Procedures: SOPs should be written in a concise, step-by-step, easy-to-read format. The information presented should be unambiguous and not overly complicated. The active voice and present verb tense should be used. The term "you" should not be used, but implied. The document should not be wordy, redundant, or overly lengthy. Keep it simple and short. Information should be conveyed clearly and explicitly to remove any doubt as to what is required. Also, use a flow chart to illustrate the process being described. In addition, follow the style guide used by your organization, e.g., font size and margins. To write a Standard Operating Procedures —»
The active voice and present verb tense should be used. The term "you" should not be used, but implied. The document should not be wordy, redundant, or overly lengthy. Keep it simple and short. Information should be conveyed clearly and explicitly to remove any doubt as to what is required. Also, use a flow chart to illustrate the process being described. In addition, follow the style guide used by your organization, e.g., font size and margins. To write a Standard Operating Procedures —»
‹•› Select Operation that is easily to manufacture, low labor cost, and consistencies in quality + manuacturing, + testing times. "What are you looking to achieve?"
‹•› Record Data of Operation methods include —» video recording, interview, flow process charts or historical records. When recording the procedure, direct observation is prefered.
‹•› Analyse the Operation —» gather all the relevant documents + observe operation being performed + ask for the technicians inputs, and take rough notes. "Colaborative Creation".
SPONSORED CONTENT

Your Service Flyer

Your Event Invitation
Your Promotion Ads

Your Logo & Brand
Support us and grow your business with us. My goal was to make technical information available with ready access to commonly needed resources, formulas, and reference materials while performing my work as a Technical Support Engineer. The businesses listed in Sponsored Content were carefully selected because of their uniqueness. However, non-sponsored selected ads will be rotated monthly.
ViBa Direct lacks an advisory board to do research and hire writers with the latest technical knowledge. Creating an effective advisory board requires more than an invitation. Without your sponsorship, this is not possible. If your company is interested in placing the company’s logo, brand, event invitation, and other promotional banners and flyers here or on any other pages, please reach out to Customer Service for more detail.
‹•› Develop the Best Method important key questions include: What tasks do we need to do? —» Who needs to do them —» What are the best ways to approach these task? —» Can the operators follow the Standard Operation safely? "Map a Process Flow Diagram".
Confirming the Steps should be carried out in the production environment where possible as a complete operation of how a prototype will look, feel and work in the real world —» "Identify Main Steps": A task Title, this will allow the task to be broken down into smaller sections. —» "Identify Key Points": Key Points are part of the operation, relevant information/steps which could impact on QUALITY–SAFETY or DELIVERY. Points of which would seriously affect the production or the success of operation.
SOPs should be written with sufficient detail so that someone with limited experience with or knowledge of the procedure, but with a basic understanding, can successfully reproduce the procedure when unsupervised. The experience requirement for performing an activity should be noted in the section on personnel qualifications. For example, if a basic chemistry or biological course experience or additional training is required that requirement should be indicated.
PREPARE THE SPECIFIC TASK
Prepare the Specific Task of the Procedure.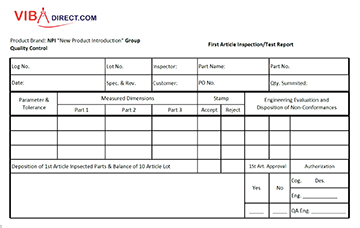 The typical approach to a document like this is to include a cover page with the title and all the relevant reference details, followed by a list of chapters, before embarking on the processes themselves. This approach is founded on common sense structure and it’s a good idea to follow it. If you’re forming a set of standard operating procedures for a particular part or feature of a content manufacturing team’s work, focusing should be on them and their needs only. Draw the line to stop wandering off into other teams or departments.
The typical approach to a document like this is to include a cover page with the title and all the relevant reference details, followed by a list of chapters, before embarking on the processes themselves. This approach is founded on common sense structure and it’s a good idea to follow it. If you’re forming a set of standard operating procedures for a particular part or feature of a content manufacturing team’s work, focusing should be on them and their needs only. Draw the line to stop wandering off into other teams or departments.
Consisting Style/Format: Be concise, be clear and communicate the important information only and don't include anything un-important or un-useful in a SOP document. Start with action commands. Always use a verb at the beginning of a statement for a task. This kind of language makes it clear what you have to do and it packs a punch. when listing detailed instructions for a particular task within the workflow, put the actionable sections first and follow with the explanation. Don’t make readers sift through paragraphs of text every time they want to follow the SOPs.
The Necessay Steps of the Process: This extra detail is important to make an easy to follow process. Collaboratively walk through the process from start to finish noting down every step which must be taken along the way. Allow for input and discussion across the team, and record down any further steps which are suggested. Go through and look at each recorded task to see if sub-task can be added to further explain either how it is performed or done productively.
Checklists: Use checklists to ensure that steps are followed in order. Checklists are also used to document completed actions. Any checklists or forms included as part of an activity should be referenced at the points in the procedure where they are to be used and then attached to the SOP. Create sub-checklists in tasks to tackle this need. Sub-checklists provide a simple way to make processes appear straightforward while also adding actionable detail. It's not good enough to assum the process step is simply working. We need to understand how it is performing so that we can optimize it.
Testing the Process: One of the methods used to test how easy it was to follow was to put new employees with the tasks and a process. Someone who was not involved in the collaborative creation process, and can look at the flows with fresh eyes. If the process was good enough, the job would be done well regardless of experience. Optimizing the process over time is a crucial step of creating it in the first place. No process is ever perfect, and nor can it be. The processes can, however, be as good as we can possibly make them. A simple checklist like below can help direct the process for optimizing a process:
‹•› Noted down which process needs optimizing —» Identify the elements of the process —» Reexamine the methods currently used —» Remove non-essential/unneccessary steps —» Noted down which steps can be automated —» Don't overlook safety in favor of speed.
It is important to remember that the processes’ strengths and weaknesses might be found by the worker who follow them each day. Stay collaborative in the optimization process. The key steps to improving the process is to consider integrating other tools and automated components into your workflow.
Create a Flow Diagram: Workflow map can be useful when giving complex instructions or explaining a process. It simplifies the process and the understanding of the instructions.
SOP Review and Approval: SOPs should be reviewed/validated by one or more individuals with appropriate training and experience with the process. The review process should not be overly cumbersome to encourage timely review. The frequency of review should be indicated by management in the organization’s Quality EPA QA/G-6 3 April 2007 Management Plan. That plan should also indicate the individual(s) responsible for ensuring that SOPs are current. It is especially helpful if draft SOPs are actually tested by individuals other than the original writer before the SOPs are finalized. The finalized SOPs should be approved as described in the organization’s Quality Management Plan. Generally the immediate supervisor, such as a section or branch chief, and the organization’s quality assurance officer review and approve each SOP. Signature approval indicates that an SOP has been both reviewed and approved by management.
NỘI DUNG TÀI TRỢ

Quảng Cáo Dịch Vụ

Quảng Cáo Sự Kiện

Quảng Cáo Khuyến Mãi

Biểu Trưng & Nhãn hiệu
Hỗ trợ chúng tôi và phát triển doanh nghiệp của bạn với chúng tôi. Mục tiêu của tôi là cung cấp thông tin kỹ thuật với khả năng truy cập sẵn sàng vào các tài nguyên, công thức và tài liệu tham khảo thường cần thiết trong khi thực hiện công việc của mình với tư cách là Kỹ sư hỗ trợ kỹ thuật. Các doanh nghiệp được liệt kê trong Nội dung được Tài trợ đã được lựa chọn cẩn thận vì tính độc đáo của chúng. Tuy nhiên, các quảng cáo liệt kê không được tài trợ sẽ được luân chuyển thay đổi hàng tháng.
ViBa Direct thiếu một ban cố vấn để thực hiện nghiên cứu và thuê các nhà văn với kiến thức kỹ thuật hiện đại. Việc tạo ra một ban cố vấn hiệu quả đòi hỏi nhiều hơn là một lời mời. Nếu không có sự tài trợ của bạn, điều này khó có thể thực hiện. Nếu công ty của bạn có nhu cầu quảng cáo, đặt biểu trưng, thương hiệu, biểu ngữ mời tham gia thảnh viên, hội viên cũng như các bích chương quảng cáo ở đây hoặc trên bất kỳ trang nào khác, xin vui lòng liên hệ với nhóm Dịch Vụ Khách Hàng Dịch Vụ Khách Hàng để biết thêm chi tiết.
Revision Update: SOPs need to remain current to be useful. Therefore, whenever procedures are changed, SOPs should be updated and re-approved. If desired, modify only the pertinent section of an SOP and indicate the change date/revision number for that section in the Table of Contents and the document control notation. SOPs should be also systematically reviewed on a periodic basis, e.g. every 1-2 years, to ensure that the policies and procedures remain current and appropriate, or to determine whether the SOPs are even needed. The review date should be added to each SOP that has been reviewed. If an SOP describes a process that is no longer followed, it should be withdrawn from the current file and archived.
Document Control: Develop a numbering system to systematically identify and label the SOP documents, and the document control should be described in its Quality Management Plan. Generally, each page of an SOP should have control documentation notation, similar to that illustrated OMS. A short title and identification (ID) number can serve as a reference designation. The revision number and date are very useful in identifying the SOP in use when reviewing historical data and is critical when the need for evidentiary records is involved and when the activity is being reviewed. When the number of pages is indicated, the user can quickly check if the SOP is complete. Generally this type of document control notation is located in the upper right-hand corner of each document page following the title page. A Mastes List of all SOPs should be created and maintain in company database server. This file should indicate the SOP number, version number, date of issuance, title, author, status, organizational division, branch, section, and any historical information regarding past versions.
Your Support Can Help Us Grow. Thank you for visiting ViBa Direct. As part of our commitment to continually improving the visitor experience. I am writing to ask you for your support, such as listing your company here with us. Clearly there is a great deal of manufacturers and distributors including both mechanical and electrical. Unfortunately, I don't have enough manpower to meet everyone needs. But if you are interested in finding out how to support us. Please provide details about your products and reach out to Customer Service, as well as any offerings that you feel may meet customer needs. Thank you in advance for taking the time to read this. Feel free to contact us with any additional feedback you would like to share about your experience visiting ViBaDirect.com.




