HOW IT WORKS?
POWDER COATING is a dry finishing process. Powder coating is a high-quality finish found on thousands of products. Powder coating protects the roughest, toughest machinery as well as the household items we depend on daily. It provides a more durable finish than liquid paints can offer, while still providing an attractive finish. Powder coated products are more resistant to diminished coating quality as a result of impact, moisture, chemicals, ultraviolet light, and other extreme weather conditions. In turn, this reduces the risk of scratches, chipping, abrasions, corrosion, fading, and other wear issues. Powder coatings are based on polymer resin systems, combined with curatives, pigments, leveling agents, flow modifiers, and other additives.
Powder coating can be accomplished through one of two processes: Thermosets and Thermoplastics. Thermosetting involves additional chemicals which react to the powder during heating. Thermoplastics have no additional chemicals and instead just melt and flow into the final coating. Both processes of powder coating look quite similar except for this distinguishing feature. The thermoset or thermoplastic powder is created by taking the powder input and binding the items together, heating the mixture, rolling out the product, and then breaking the polymer product into chips which can be ground into a fine powder.

BBVA Bancomer Stadium "El Gigante de Acero" developed by FEMSA & C.F. Monterey in Guadalupe, Mexico
FUNDAMENTAL OF POWDER COATING. Electrocoating is a process by which electrically charged particles are deposited out of a water suspension to coat a conductive part. During the electrocoat process, paint is applied to a part at a certain film thickness, which is regulated by the amount of voltage applied. The deposition is self-limiting and slows down as the applied coating electrically insulates the part. Electrocoat solids initially deposit in the areas closest to the counter electrode and, as these areas become insulated to current, solids are forced into more recessed bare metal areas to provide complete coverage. This phenomenon is known as throwpower and is a critical aspect of the electrocoat process.
WHY POWDER COATING? More and more engineers have turned to powder coating as a way to produce a high-quality finish while increasing production, cutting costs, and complying with increasing environmental pressure. The cost efficiencies in electrocoat are higher transfer efficiency, precise film-build control, and low manpower requirements. Increased line productivity in electrocoat is due to faster line speeds, dense racking of parts, non-uniform line loading, and reduced human fatigue or error. The environmental advantages are no- or low-VOC and HAPs products, heavy metal-free products, reduced exposure of workers to hazardous materials, reduced fire hazards, and minimum waste discharge.
 RESERVED FOR ⭐ SPONSORS
RESERVED FOR ⭐ SPONSORS

BRAND VISIBILITY. If your company is interested, please contact for more detail.

AUDIENCE REACH. If your company is interested, please contact for more detail.

DRIVE SALES. If your company is interested, please contact for more detail.
— ECONOMIC CONSIDERATION. The excellence of the powder-coated finish is accompanied by substantial cost savings, when compared to liquid coating systems. Since powder contains no VOCs, air used to exhaust the powder spray booth can be recirculated directly to the plant, eliminating the cost of heating or cooling the makeup air. Ovens that cure solventbased coatings must heat and exhaust huge volumes of air to ensure that the solvent fumes do not reach a potentially explosive level. With no solvent in powder coating, the exhaust required in the ovens is lower, resulting in energy and cost savings in spite of the higher curing temperatures ~ that powder coating requires.
— ADVANTAGE OF POWDER COATING OVER PAINT. Powder coating results in a thick, dense finish on metal products which can be more durable and longer lasting than conventional painting. A powder coated surface will be more resistant against scratches, chipping, wear, and fading compared to other type of finishes. When powder coated items are installed without damage to the powder coating and they are maintained regularly, they should be relatively permanent. The correctly applied coating, although not metallurgically bonded to the metal will not crack, chip or peel as with conventional paint films. It is resistant to cracking, peeling, chipping, abrasion, rust and damage due to chemical exposure. While it isn't indestructible, it is highly durable, retains colour and gloss, goes on uniformly, has excellent UV stability and is a far more environmentally friendly option than traditional liquid paint. Liquid painting and powder coating are two techniques used in industrial finishing. Both typically require a heat curing process, but they differ greatly in their methods of application. The paint finish differs according to the surface being painted.
POWDER COATING PROCESS
POWDER COATING PROCESS. The powder coating process is very similar to a painting process except that the “paint” is a dry powder rather than a liquid. The finely ground particles of pigment and resin are electrostatically charged and sprayed onto a metal part. Any substrate can be used that can tolerate the heat of curing the powder and that can be electrically grounded to enhance charged particle attachment. The powder flows and cures during the application of heat. After application of the powder coating, the parts enter a curing oven where, with the addition of heat, the coating chemically reacts to produce long molecular chains, resulting in high cross-link density. These molecular chains are very resistant to breakdown. The result is a uniform, durable, high-quality finish. This type of application is the most common method of applying powders. Powder coatings can also be applied to non-metallic substrates such as plastics and medium density fiberboard (MDF).
A typical steps in a powder coating process for metal part is illustrated below...
1. PRE-TREATMENT/CLEANING. In the typical metal finishing process, the alkaline cleaning tanks are first in line and take the bulk of the dirt load. Whether the tanks are soak cleaners, ultrasonic assisted cleaners, or electrocleaners, their purpose is to remove oils, grease, wax, polishing compound, particulates, and light oxides from the part surfaces. Depending on the detergent additives in these tanks, the tanks could build up surface oil, oil emulsions, suspended solids, or sludge at the bottom of the tank or any combination of these contaminants. As with acids, the cleaning chemicals are consumed in the process of removing and preventing redeposition of the contaminants.
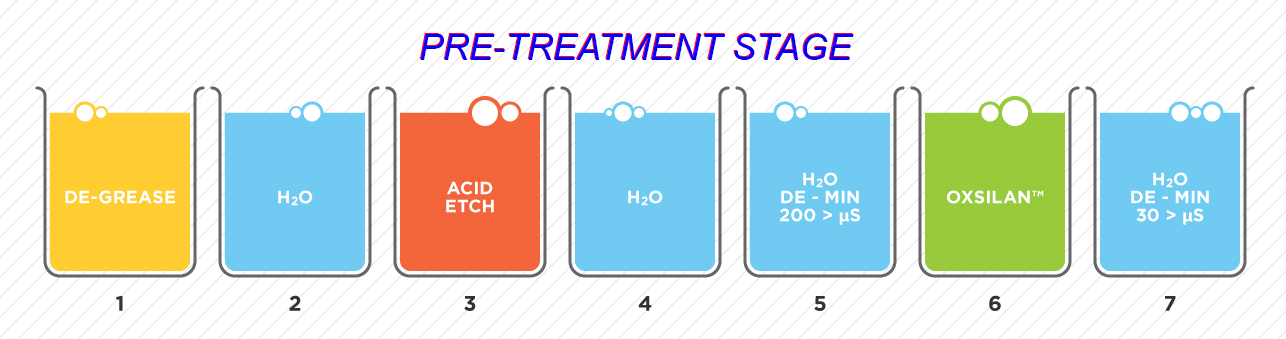
2. RINSING. Rinsing is critical in the metal finishing process but more water use does not necessarily mean better rinsing. Best practices for producing effective rinsing are: Spray rinsing – Rinse controls such as automatic valves controlled by timers or water conductivity limits. – Reactive rinsing for the appropriate process chemistry combinations. – Double counterflow immersion rinse tanks between process tanks.
3. PHOSPHATING. The process of phosphating aluminum and steel parts is typically listed as a conversion coating because the process involves metal removal as part of the reaction. However, it is not like anodizing or black oxide in that the phosphate coating is actually a precipitation reaction. The final surface is a layer of very fine phosphate crystals adhering to the surface of the metal.
For paint and powder coatings, a phosphate coating has two main functions. First, the coating provides improved paint and powder coating adhesion since the phosphate crystals act as organic coating anchoring sites. Second, the phosphate layer acts as a corrosion barrier should the organic coating get scratched. In rust creep testing, the rust creep is reduced when phosphate is present under the paint layer or powder coat layer compared to no conversion layer under the organic coating. The most common phosphating chemistries are iron phosphate, zinc phosphate, and manganese phosphate. Phosphate can be used as a stand-alone coating for other purposes such as lubricity in parts forming. Most phosphating lines are single tank three step, or five step processes.
The three and five stage processes are:
- Three Stage Process: Clean–Phosphate — » Rinse — » Rinse–Seal.
- Five Stage Process: Clean — » Rinse — » Activate Rinse — » Phosphate — » Seal.
4. DRYING. The energy intensive operations in the powder coating operation are drying and curing. Typical drying and curing operations are with the use of convection ovens. Drying to prevent rusting before some additional steps, to avoid water spots resulting in some cosmetic problems, or is it to provide dry parts for the next operation? The easiest way to prevent water spotting is with the use of DI (deionized) water in the final rinse. The water spots are typically due to minerals in normal process water and can be eliminated by removal of the minerals.
Another method that at least will minimize water spots is with an air blow-off process to remove the water droplets before they have time to dry on the parts. This method will be very dependent on part geometry and part orientation on the racks. If the air cannot reach certain areas on the parts due to geometry or if parts block each other then the spotting may still occur in those areas. high pressure blowers should be used instead of compressed air.
5. COATING. After pre-treatment the part is suspended on a conveyorised production line and passes into the spray booth for coating. The powder which is held in a fluidised hopper bed is carried to the spray gun using compressed air. As the powder passes out of the gun, the particles are charged and therefore attracted to the earthed piece of metal. This creates an even coating as it wraps around the work piece.
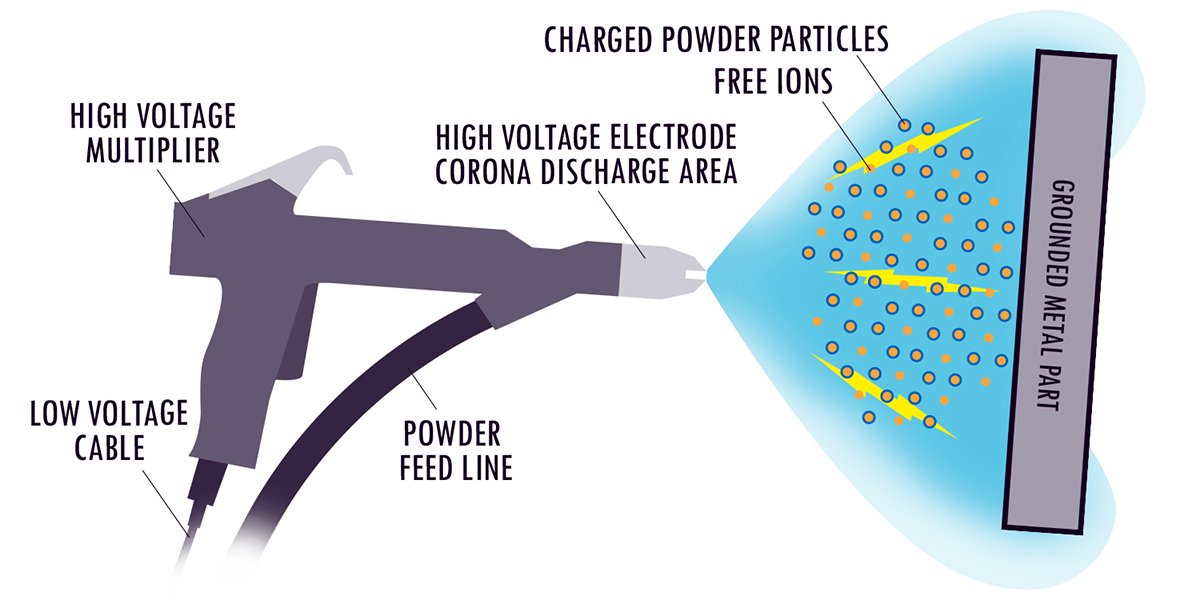
6. CURING. Depending on the part geometry, simple geometries can be dried and cured using infra red heating. The heating occurs at the part surface with little air heating of bulk heating of the part. Interesting side note, the use of infra red curing for liquid paints has an interesting and helpful effect. The infrared light heats the part surface and not the paint, resulting in paint drying from the inside out. The paint does not skin over and trap solvent or water inside the paint as can sometimes happen with convection curing. 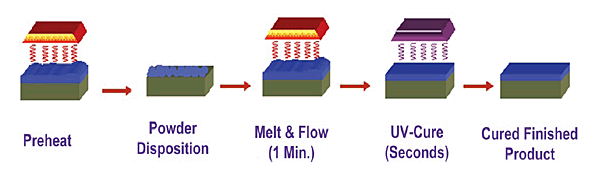 All the volatiles are driven out of the paint, starting at the part surface and progressing to the paint surface. Ones might say that the paint dries backwards.Therefore the heat energy is going where it is needed, to the surface. It is also a very rapid heating process, again since there is instant infra red light absorption.
All the volatiles are driven out of the paint, starting at the part surface and progressing to the paint surface. Ones might say that the paint dries backwards.Therefore the heat energy is going where it is needed, to the surface. It is also a very rapid heating process, again since there is instant infra red light absorption.
Another advantage of infra red heating is in the powder coat curing process. The rapid part surface heating results in better powder flow and less chance of dust or dirt defects since there is very little air flow to deposit particles compared to convection heating.
 RESERVED FOR ⭐ SPONSORS
RESERVED FOR ⭐ SPONSORS

BRAND VISIBILITY. If your company is interested, please contact for more detail.

AUDIENCE REACH. If your company is interested, please contact for more detail.

DRIVE SALES. If your company is interested, please contact for more detail.
How Long Does It Take for Powder Coating to cure? Depending on the metal thickness determines the amount of time the piece needs to “bake”. The average temperature is 400 degrees for approximately 30 minutes. The curing schedule can also vary depending on the manufacturer's powder specifications. To cure powder coating, both the powder and the substrate have to reach the desired temperature and stay there for the allotted time without any temperature fluctuations. For this reason, the best way to cure powder coating is with a curing oven.
THE DRAWBACKS. There are two major drawbacks to the powder coating process. First, powder coating produces a thick finish to metal products. It can be quite difficult for powder coating to produce thin finishes, particularly because as the polymer is thinned it tends to produce a bumpy texture similar to the peel of an orange. Second, smaller sheet metal jobs might prefer a less expensive or complex finishing process. Powder coating requires spray materials, electrostatic booth, and an oven—all items which can be expensive and involved for smaller projects.
The benefits of wet paint and plating are quite complimentary to powder coating. First, wet paint is ideal for products which cannot be heated for powder coating because wet paint does not require an oven for finishing. Second, wet paint can produce a wider range of colors than powder coating so more custom color work could require spray painting and plating. Third, wet paint and plating can produce a much thinner finish than powder coating. Products with demand a thin finish can benefit greatly from wet paint. Finally, wet paint is a much more economic finishing process, particularly for smaller jobs.
